US Pharmaceutical Company Tonix to Conduct Monkeypox Vaccine Trials in Kenya
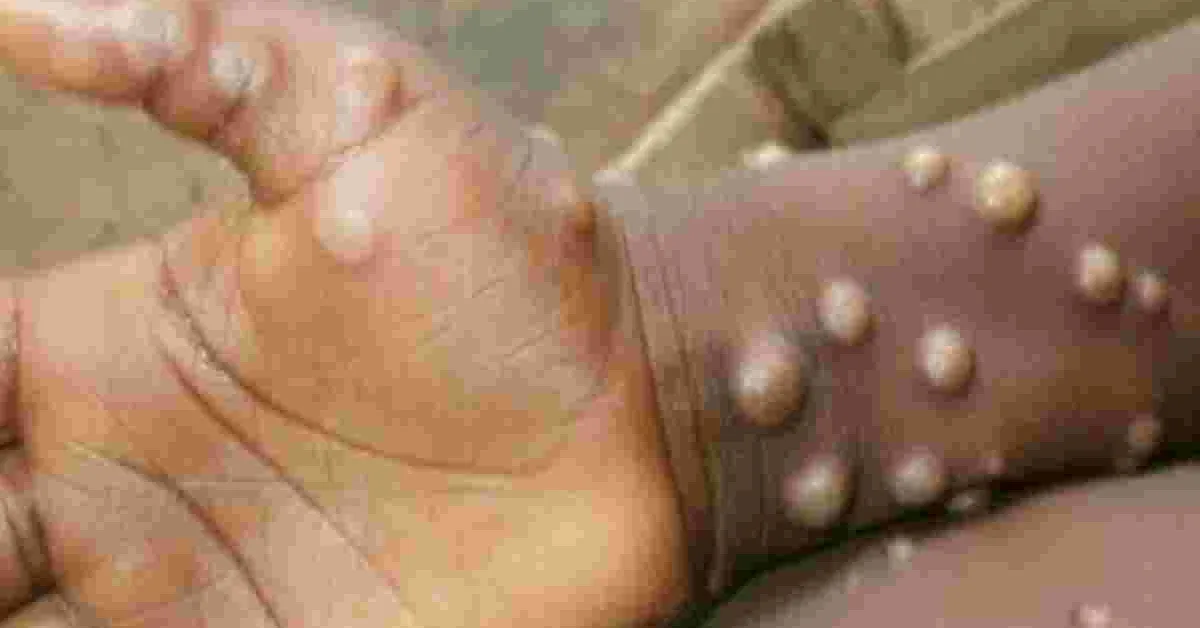
US pharmaceutical company Toxin has picked Kenya for a pilot study and vaccine trial for monkeypox and smallpox.
Tonix Pharmaceuticals Holding Corp, a clinical-stage biopharmaceutical company, will partner with the Kenya Medical Research Institute (KEMRI) for the study that will commence in the first half of 2023.
In a statement, the company said it will collaborate with KEMRI to plan, seek regulatory approval and conduct a Phase 1 clinical study in Kenya to develop TNX-8011 as a vaccine to protect against monkeypox and smallpox.
“KEMRI is excited to plan this clinical trial with Tonix, and ultimately to lead the trial. Monkeypox has spread in Central and West Africa, and there’s a concern that we could begin seeing cases in the Eastern and Central Africa or from foreign travelers,” KEMRI Director General and CEO Prof. Samuel Kariuki said on Monday.
Toxin CEO Dr. Seth Lederman said Phase I of the study will begin in the first half of 2023, with KEMRI leading the study and Tonix supporting regulatory filings and other logistics.
Kenya has no reported cases of Monkeypox which is spreading around the world, with at least 16,000 infections documented in more than 40 countries. The disease is caused by infection with monkeypox virus, a virus closely related to variola virus that causes smallpox.
The World Health Organisation (WHO) has declared Monkeypox a public health emergency of international concern, with scientists indicating that it spreads mainly through sexual experiences.
The global outbreak of monkeypox surfaced in the United Kingdom in May. Cameroon, Nigeria, and the Central African Republic are the only African nations that have recorded cases of the virus so far.














Comments
Do a clinical trial on your…
Permalink
Do a clinical trial on your own people or pick a European country.
Why in Kenya if the disease…
Permalink
Why in Kenya if the disease is spreading in: UK, US, Denmark, Canada, Germany, Central Africa, Australia, West Africa,Spain- which is said to be world's worst hit by the disease. 70% of those cases are found in Europe! Kenya has zero cases right now. Are they going to bring it in Kenya? What else will they test and bring to our people under the guise of these trials? Buyer be aware!
Don't worry, somebody on the…
Permalink
In reply to Why in Kenya if the disease… by Mwororo (not verified)
Don't worry, somebody on the top got a beautiful paycheck and all we see is downstream thieves. offcourse they did not steal, they got a kickback. Muichi amurikaga uria ungi.
Don’t worry, they’ll make…
Permalink
In reply to Why in Kenya if the disease… by Mwororo (not verified)
Don’t worry, they’ll make sure to bring the disease here in kenya first.
" US pharmaceutical company"…
Permalink
" US pharmaceutical company"?
Why are they conducting trials while there are no monkeypox cases in Kenya?
Can Essau be trusted to conduct fair trials using a black man? ( In Kenya).
The TUSKEGEE study of untreated Syphilis in African American male (blacks) was conducted between 1932 and 1972 by the United States Public Health service (PHS) and Centers for Disease control using a total of 600 impoverished African American share cropper from Macon County, Alabama.
Initially, the experiment was only going to take six months but was extended to 40 years until 1972 when a leak to the press forced it to stop.
The sick African American men were never treated with penicillin despite the fact that by 1947, the antibiotic was widely available and was already being used to treat white people.
Blacks who were sick were left to die, and their wives and children were left sick with the disease.
Kenyans, Just remember what white people are capable of doing to black people!!
Soon there will be alot of…
Permalink
Soon there will be alot of cases monkey fox in that negro land
Only foolish low-IQ corrupt…
Permalink
Only foolish low-IQ corrupt African leaders and research scientists will approve this. They know very well this cannot be allowed in the US. They also know that they cannot do this directly but indirectly by dishing out millions of cash to Uhuru and Raila to support their campaign. Soon, expect Mutahi Kagwe to give a ministry of health response to this.Uhuru doesn't care about citizens because he conned them to vote for him but now he is treating them like mafi ya kuku. What a dishonest way to thank those who voted for him? Tukutane kwa debe.
🛑✋🏿🛑✋🏿. Wait a freeeaaking…
Permalink
🛑✋🏿🛑✋🏿. Wait a freeeaaking minute, monkeypox vac’s flying off the shelves over here like there’s no tomorrow and you over there looking for lab monkeys. This shit rediculous, try West Africa where the apparent outbreak originated. Jinga type
Africans exposed to the…
Permalink
Africans exposed to the disease then make trials on them, why didnt thy do it in USA??....Who's are these individuals signing these contracts???....KEMRI blackmailed with cash!..
We do not have monkeypox in…
Permalink
We do not have monkeypox in Kenya. So, do it in your own countries.
Leave Kenya alone if your…
Permalink
Leave Kenya alone if your motive is genuine you go to where to those most affected live who will gladly volunteer to be tested and treated of this disease.Start in the USA furthermore charity starts at home and then proceed to central and west Africa if you “have” to be in Africa so as to fake that you give a rats ass about what goes on in this continent.
History has taught Africans…
Permalink
History has taught Africans nothing.
Aren't these the same companies who were infecting African Americans with STDS for clinical trials?
How comes Monkeys pox is affecting the WEST now?
Anyway, all our corrupt officials are seeing right now are the $$$ signs with no regard to human life.
If there are no known cases…
Permalink
If there are no known cases how the hell are they going to do the damn trials? bring it to kenyan people so they can do triala? I hope ruto take over the government next week and reject shyt like this. uhuru has sold us to china now giving american access to use our people as guinea pigs? wow
This is what happens when…
Permalink
This is what happens when historical evils visited upon a people are not satisfactorily solved or addressed. The victim is always suspicious of the evil doers intentions.This is a big problems because many blacks will always read malice in any project such as the one suggested here,even when no malice exists.Only full retribution will start healing the wounds that were inflicted on our forefathers,and passed on to us,their children.
My people, complain all you…
Permalink
My people, complain all you want but the facts will not change any time soon. Poor people and countries are treated like children of a lesser God the world over. Do rich criminals do prison time in Kenya? Has anyone of you ever heard of a body from a wealthy family being detained at a funeral home because of unpaid bills? Has the daughter of an affluent Kenyan been employed in Saudi as a domestic worker and ended up beaten to death? Next the Chinese will be testing their "Sinovax" vaccine that supposedly will be a cure for corruption tendencies in Kenya. Accept and move on. In the meantime, I am going to answer a call light in room 5 at my nursing home job mama mzungu anataka nimpanguze matako.
Hehehehe Blacks r cursed all…
Permalink
Hehehehe Blacks r cursed all over the world.The same way Christianity aka Jesus was pushed brainwashed in our minds n wr almost kuku following the white man n now the monkey this that so we remain the stupid folks out there.Nonsense
Stop complaining you people…
Permalink
Stop complaining you people. His excellency the former president of USA Donald Trump said . Africa is full of shit hole countries. He was very right on so many levels. Because African countries like to be treated like shit and so they are. Show me your boundaries and I will respect them, simple. Someone in Whatever KEMRI means is making big money along with the some rogue leaders. This is not the first time . I read somewhere a few years ago Kenya allowed sterilization of women through the church in name of free vaccines. And now this . I’m sure there another one on the way and Kenyans will do nothing about it. They will just whine online and then shut up. The project will continue as planned and there nothing they can about it. More corporations are on the way to utilize the free availability of laxities and vulnerability of the Kenyans and Africans in general. You accept it without complaining coz it is what it is. Tonix did not force anyone
Add new comment